about us
Neuheit Research
(Neuheit Services LLP)
We partner with pharmaceutical companies, biotechnology firms, investigators, hospitals, and research institutions to support end-to-end execution of clinical trials across diverse therapeutic domains.
- Comprehensive Clinical Trial Management
- Regulatory & Ethical Compliance
- Experienced Scientific & Medical Team
Our Services
At Neuheit Research, we provide end-to-end clinical research services, combining deep expertise, advanced technologies, and regulatory excellence to support sponsors throughout the lifecycle of clinical trials. Our multidisciplinary team ensures quality, compliance, and efficiency, enabling sponsors to achieve trial objectives confidently.

Clinical Operations
Effective clinical operations are the backbone of successful clinical research.

Clinical Data Management
Accurate, secure, and compliant data is the foundation of successful clinical research.

Regulatory Affairs Services
Navigating regulatory requirements is critical for the successful conduct of clinical trials.

Pharmacovigilance
Protecting patients, ensuring compliance, and enabling confident decision-making—this is the essence of our Pharmacovigilance services.

Quality Assurance
Ensuring compliance, accuracy, and integrity is the cornerstone of Neuheit Research’s Quality Assurance services.

Remote Monitoring Services
Clinical trials involve multiple critical processes that require careful oversight.

Medical Writing Services
High-quality, clear, and regulatory-compliant documentation is essential for successful clinical research.
Why Choose Neuheit Research
Trusted Partner in Ethical & High-Quality Clinical Research
End-to-End Clinical Trial Expertise
Patient-Centric & Ethical Approach
Strong Quality & Regulatory Framework
Integrity-Driven Collaboration

16 +
1 k
172
112 +
Our Therapeutic Focus
Therapeutic Areas
of Expertise
We provide comprehensive clinical research support across multiple therapeutic domains, addressing disease-specific challenges with scientific precision, regulatory compliance, and a patient-centric approach.
Our Clinical Trial Execution Framework

Define the Study
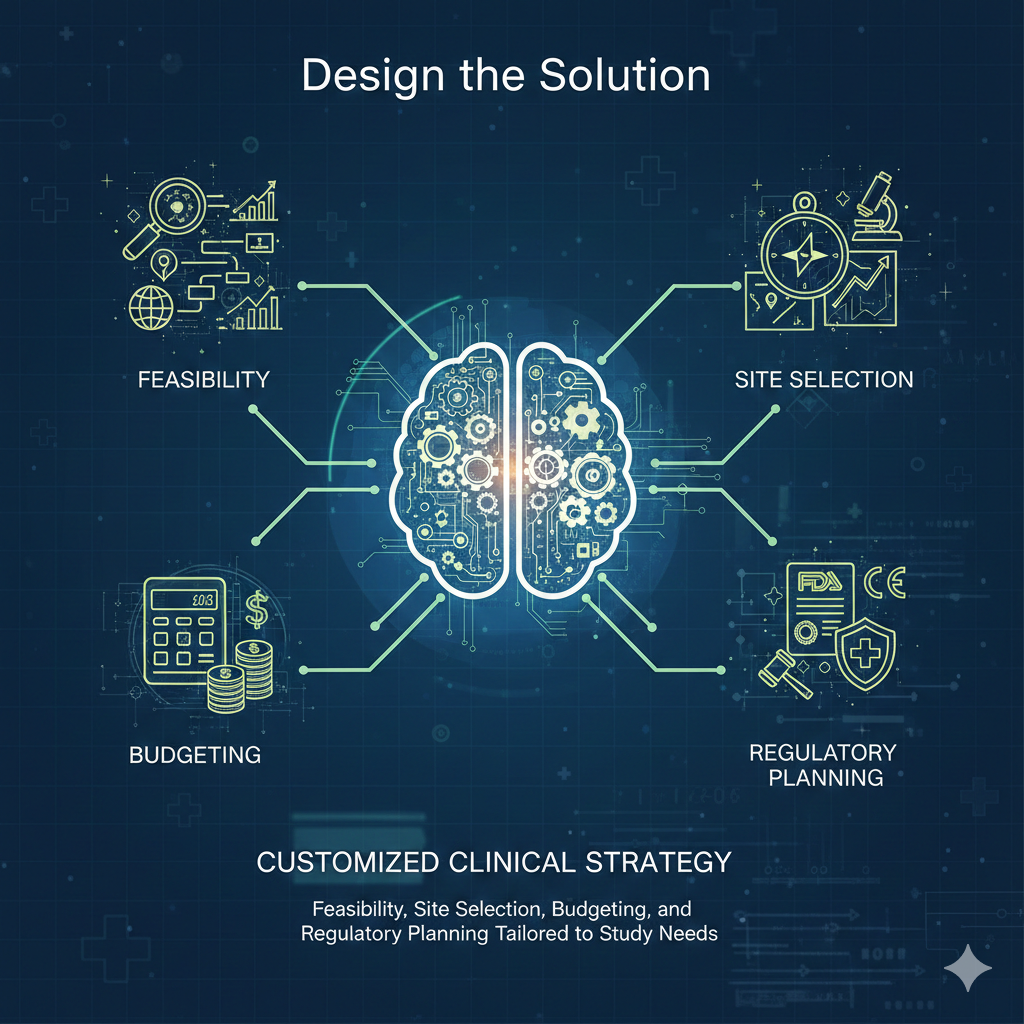
Design the Solution

Execute the Trial

Deliver Results
testimonials
Some feedbacks from
honorable clients
700 +
Dr Sanjay Mankar
We recently completed a study on antibiotic-associated diarrhea at Mankar Hospital in Pune. From the outset, Neuheit demonstrated a clear understanding of our study goals and worked closely with us to ensure that the study was conducted with the highest level of quality and accuracy.

Team member
Meet our professional
science specialists

Dr Souraabh S Ambapkar

Dr. Subhal B Dixit

Dr Saanvi S Ambapkar

Dr. Tushar Ranaware
about us
A fair company for laboratory products
Certified laboratory of world health organization
Best laboratory award winner year 2016-2017
Client 01

Client 06

Client 05

Client 04

Client 03

Client 02

Client 01